 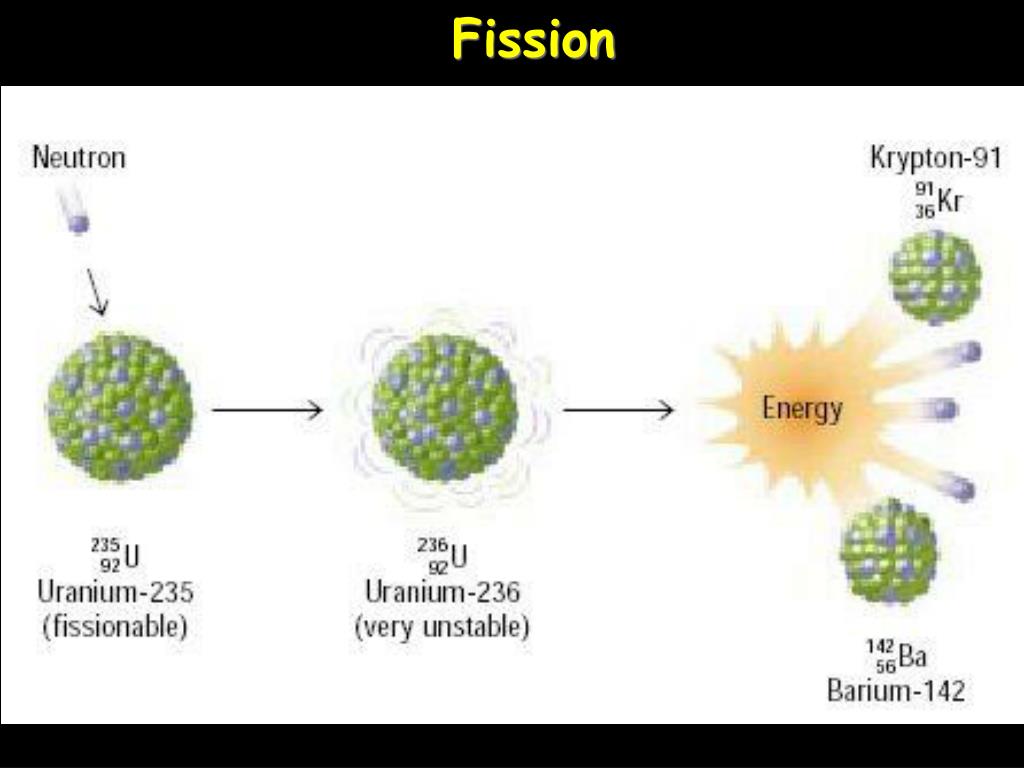  
The fission of weighty components is profoundly exothermic which discharges. Due to the much larger energy differences between nuclear energy shells, gamma rays emanating from a nucleus have energies that are typically millions of times larger than electromagnetic radiation emanating from electronic transitions.Ī balanced chemical reaction equation reflects the fact that during a chemical reaction, bonds break and form, and atoms are rearranged, but the total numbers of atoms of each element are conserved and do not change. Presentation: Introduce the concept of nuclear fission and explain that set up on the cart is an example of a nuclear reaction containment vessel uncover so. Fission is the parting of a nucleus that delivers free neutrons and lighter nuclei. Young University of Illinois at Chicago via The process by which nitrogen is converted to carbon-14 is an example of neutron capture, in which particles are absorbed by the nucleus of another atom to form a new element. Because the sum of the mass numbers of the reactants must equal the sum of the mass numbers of the products: 25 + 4 A + 1. Z is the atomic number of the new nuclide, X. Gamma rays are produced when a nucleus undergoes a transition from a higher to a lower energy state, similar to how a photon is produced by an electronic transition from a higher to a lower energy level. 11.6: Nuclear Fission Last updated 11.5: Radioactive Half-Life 11.7: Nuclear Fusion Paul R. Nuclear Fission Isotopes of uranium and plutonium both undergo fission and are used as fuels in nuclear power stations During fission, when a neutron collides. The nuclear reaction can be written as: Mg25 12 + He4 2 H1 1 + XA Z. Gamma rays compose short wavelength, high-energy electromagnetic radiation and are (much) more energetic than better-known X-rays. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
